2017-05-08 15:16来源:互联网作者:上海管理员
摘要:在某些溶液中,甜菜细胞经历薄膜破裂,使细胞释放出甜菜花青苷(一种红色的色素)。AS薄膜破裂越多,更多的甜菜花青苷被释放出来。我们通过计算被溶液吸收的光量(在某一个特定的波长)来决定甜菜花青苷释放的量。当溶液中甜菜花青苷的浓度增加时,吸收的光的量也增加。
Practice 4
(63C-Passage I)
在某些溶液中,甜菜细胞经历薄膜破裂,使细胞释放出甜菜花青苷(一种红色的色素)。AS薄膜破裂越多,更多的甜菜花青苷被释放出来。我们通过计算被溶液吸收的光量(在某一个特定的波长)来决定甜菜花青苷释放的量。当溶液中甜菜花青苷的浓度增加时,吸收的光的量也增加。
为了决定被溶液吸收的光的量,光可以直接穿过溶液的样品到达探测器。
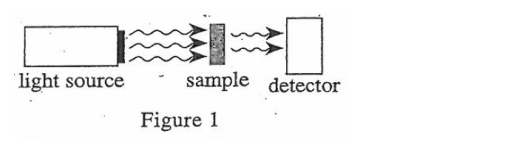
By measuring the amount of light that hits the detector, the amount of light absorbed by the sample can be determined.
Experiment 1
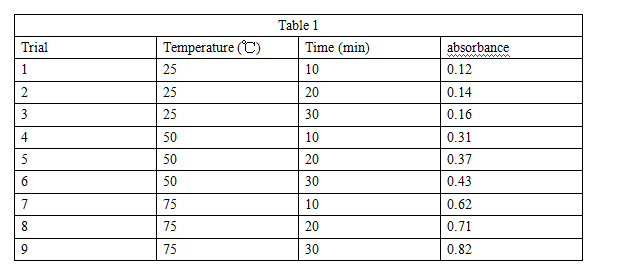
For each of 9 trials, a student placed a beet section (2 cm × 2 cm × 1 cm) in 100 mL of water at a specific temperature for 10, 20, or 30 min. The student then removed a sample of the solution and determined its absorbance (see Table 1).
Experiment 2
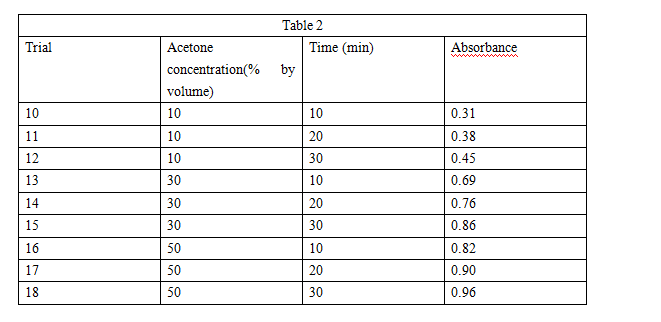
Experiment 1 was repeated, except that in each trial an acetone/water solution was used instead of water and the temperature was kept constant at 250℃ (see Table 2).
Experiment 3
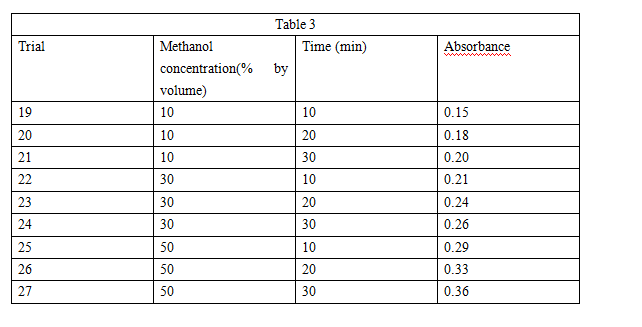
Experiment 2 was repeated, except that in each trial a methanol/ water solution was used instead of an acetone/water solution (see Table 3).
The student then determined the absorbance of water, of acetone, and of methanol. Each had an absorbance of 0.00.
Practice 4.1
Suppose that in Experiment 3 a beet section had been placed in a solution that was 40% methanol by volume for 20 min. The absorbance of the sample from the resulting solution would most likely have been closest to which of the following?
A. 0.15
B. 0.30
C. 0.45
D. 0.60
Practice 4.2
Based on the results of Experiment 2, which of the following additional trials would have resulted in an absorbance closest to 0.80?
F. 10% acetone for 15 min
G. 10% acetone for 25 min
H. 30% acetone for 25 min
J. 30% acetone for 35 min
Practice 4.3
The results of Experiment 1 are most consistent with which of the following conclusions about the effects of temperature on membrane disruption and absorbance? Higher temperatures resulted in:
A. greater membrane disruption and higher absorbances.
B. greater membrane disruption and lower absorbances.
C. less membrane disruption and higher absorbances.
D. less membrane disruption and lower absorbances.
Practice 4.4
The student concluded that at a given concentration, acetone causes more membrane disruption than does methanol. Is this conclusion supported by the results of Experiments 2 and 3?
F. No, because at each concentration tested, more betacyanin was released in the acetone solution than was released in the methanol solution.
G. No because at each concentration tested, less betacyanin was released in the acetone solution than was released in the methanol solution.
H. Yes, because at each concentration tested, more betacyanin was released in the acetone solution than was released in the methanol solution.
J. Yes, because at each concentration tested, less betacyanin was re eased in the acetone solution than was released in the methanol solution.
Practice 4.5
Suppose that in Experiment 1, betacyanin concentration was directly proportional to absorbance. Accordingly, the betacyanin concentration in the sample in Trial 7 was most likely twice as great as the betacyanin concentration in the sample in:
A. Trial 3.
B. Trial 4.
C. Trial 5.
D. Trial 6.
Practice 4.6
In Experiment 1, the student directly varied 2 independent variables and measured how these changes affected the value of the dependent variable. Which of the following lists an independent variable and the dependent variable in Experiment 1?
Independent variable _______________ Dependent variable
F. absorbance water _______________ concentration
G. water concentration methanol______ concentration
H. methanol concentration ____________ temperature
J. temperature _____________________ absorbance
Answers for Practice: Practice 4 B H A H B J
上海新航道学校作为业内良心ACT培训机构 ,坚持“英语”的理念,注重学术研究,注重培养学生的学习能力。想成为下一个高分学员?赶紧来新航道报名ACT培训班 吧!
免费领取最新剑桥雅思、TPO、SAT真题、百人留学备考群,名师答疑,助教监督,分享最新资讯,领取独家资料。
方法1:扫码添加新航道老师

微信号:shnc_2018
方法2:留下表单信息,老师会及时与您联系
免责声明
1、如转载本网原创文章,请表明出处;
2、本网转载媒体稿件旨在传播更多有益信息,并不代表同意该观点,本网不承担稿件侵权行为的连带责任;
3、如本网转载稿、资料分享涉及版权等问题,请作者见稿后速与新航道联系(电话:021-64380066),我们会第一时间删除。
地址:徐汇区文定路209号宝地文定商务中心1楼
乘车路线:地铁1/4号线上海体育馆、3/9号线宜山路站、11号线上海游泳馆站
总部地址:北京市海淀区中关村大街28-1号6层601 集团客服电话:400-097-9266 总部:北京新航道教育文化发展有限责任公司
Copyright © www.xhd.cn All Rights Reserved 京ICP备05069206

 微信公众号
微信公众号
 微信社群
微信社群