摘要:今天继续分享75道经典常考的AP化学选择题2,赶紧来锻炼一下吧!答案在文章结尾。
今天继续分享75道经典常考的AP化学选择题2,赶紧来锻炼一下吧!答案在文章结尾。
Part B
Directions: Each of the questions or incomplete statements below is followed by five suggested answers or completions. Select the one that is best in each case and then fill in the corresponding oval on the answer sheet.
Questions 15-16 relate to the graph below. The graph shows the temperature of a pure substance as it is heated at a constant rate in an open vessel at 1.0 atm pressure. The substance changes from the solid to the liquid to the gas phase.
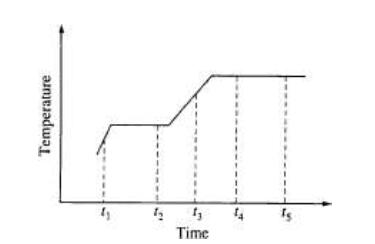
15. The substance is at its normal freezing point at time
(A) t1 (B) t2 (C) t3 (D) t4 (E) t5
16. Which of the following best describes what happens to the substance between t4 and t5?
(A) The molecules are leaving the liquid phase.
(B) The solid and liquid phases coexist in equilibrium.
(C) The vapor pressure of the substance is decreasing.
(D) The average intermolecular distance is decreasing.
(E) The temperature of the substance is increasing.
17. In which of the following groups are the three species isoelectronic; i.e., have the same number of electrons?
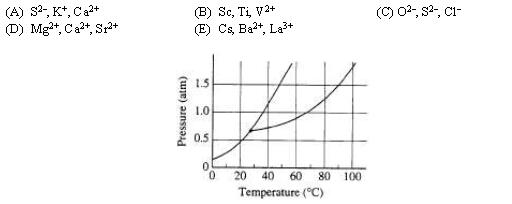
18. The phase diagram for the pure substance X is shown above. The temperature of a sample of pure solid X is slowly raised from 10˚C to 100˚C at a constant pressure of 0.5 atm. What is the expected behavior of the substance?
(A) It first melts to a liquid and then boils at about 70˚C.
(B) It first melts to a liquid and then boils at about 30˚C.
(C) It melts to a liquid at a temperature of about 20˚C and remains a liquid until the temperature is greater than 100˚C.
(D) It sublimes to vapor at an equilibrium temperature of about 20˚ C.
(E) It remains a solid until the temperature is greater than 100˚C.

答案:15B 16A 17A 18D 19E 20E 21B 22C 23D 24C 25A
更多AP化学真题及考点,请关注:https://sh.xhd.cn/apchemistry/
新航道上海AP课程培训中心,是新航道集团和美国Kaplan教育集团共建设,美国大学理事会在中国授权的AP嵌入式课程学习中心,AP Code:694387。
我们开设有多重AP课程,感兴趣的同学还可以在线咨询哦
上一篇:AP化学备考知识点十问十答
下一篇:75道经典AP化学选择题型(一)
免费领取最新剑桥雅思、TPO、SAT真题、百人留学备考群,名师答疑,助教监督,分享最新资讯,领取独家资料。
方法1:扫码添加新航道老师

微信号:shnc_2018
方法2:留下表单信息,老师会及时与您联系
| 课程名称 | 班级人数 | 课时 | 学费 | 报名 |
|---|---|---|---|---|
| AP宏观/微观经济学 | 3-6人 | 40课时 | ¥11800元 | 在线咨询 |
| AP微积分 | 3-6人 | 40课时 | ¥11800 | 在线咨询 |
| AP统计学 | 3-6人 | 40课时 | ¥11800 | 在线咨询 |
| AP计算机原理 | 3-6人 | 40课时 | ¥11800 | 在线咨询 |
| AP化学 | 3-6人 | 40课时 | ¥11800 | 在线咨询 |
| 课程名称 | 班级人数 | 课时 | 学费 | 报名 |
|---|---|---|---|---|
| AP物理1培训班 | 3-6人 | 32课时 | ¥11800 | 在线咨询 |
| AP物理2培训班 | 3-6人 | 32 | ¥11800 | 在线咨询 |
| 课程名称 | 班级人数 | 课时 | 学费 | 报名 |
|---|---|---|---|---|
| AP美国历史 | 3-6人 | 80课时 | ¥22800 | 在线咨询 |
| AP世界历史 | 3-6人 | 60课时 | ¥22800 | 在线咨询 |
| 课程名称 | 班级人数 | 课时 | 学费 | 报名 |
|---|---|---|---|---|
| AP一对一 | 1人 | 按需定制 | ¥ | 在线咨询 |
免责声明
1、如转载本网原创文章,请表明出处;
2、本网转载媒体稿件旨在传播更多有益信息,并不代表同意该观点,本网不承担稿件侵权行为的连带责任;
3、如本网转载稿、资料分享涉及版权等问题,请作者见稿后速与新航道联系(电话:021-64380066),我们会第一时间删除。